|
3/23/2023 0 Comments Blood brain barrierThe most extensively pericyte-deficient brain microvasculature compatible with adult life observed so far occurs in mice carrying a homozygous deletion of the PDGFB heparan sulfate proteoglycan-binding motif ( Pdgfb ret/ret mice). 17, 18 Pericyte-deficient adult mice can, however, be generated through partial PDGFB/PDGFRB loss-of-function approaches. PDGFB (Platelet-derived growth factor-B) signaling through PDGFRB (PDGF receptor-β) is essential for pericyte recruitment during developmental angiogenesis and postnatal survival in mice. 16 However, whether pericyte damage contributes causally, and how and to what extent pericytes regulate the functional state of ECs in the pathogenesis of these conditions, are still not clear. 3–7 Several CNS diseases have been associated with pericyte loss or damage, including diabetic retinopathy, 8–10 stroke, 11 Alzheimer disease, 12–14 amyotrophic lateral sclerosis, 15 and aging. 3–6 Developmental absence or reduction of pericytes leads to increased endothelial transcytosis and misexpression of molecular transporters and leukocyte adhesion molecules. Indeed, studies suggest that pericytes are critically required for the establishment of fully functional blood-brain barrier (BBB) and blood-retina barrier. The central nervous system (CNS) microvasculature is considered to have a high pericyte density compared with peripheral organs, 2 suggesting a specific importance of pericytes in the CNS. 1 Pericyte morphology, distribution, and density vary between organs and vascular beds. While VSMCs surround arteries and veins and, depending on vessel type, regulate vascular tone, and diameter, pericytes are typically embedded within the basement membrane of blood capillaries, where they establish direct contacts with ECs. Pericytes and vascular smooth muscle cells (VSMCs), collectively referred to as vascular mural cells, form together with the endothelial cells (ECs) the cellular component of the blood vessel wall. 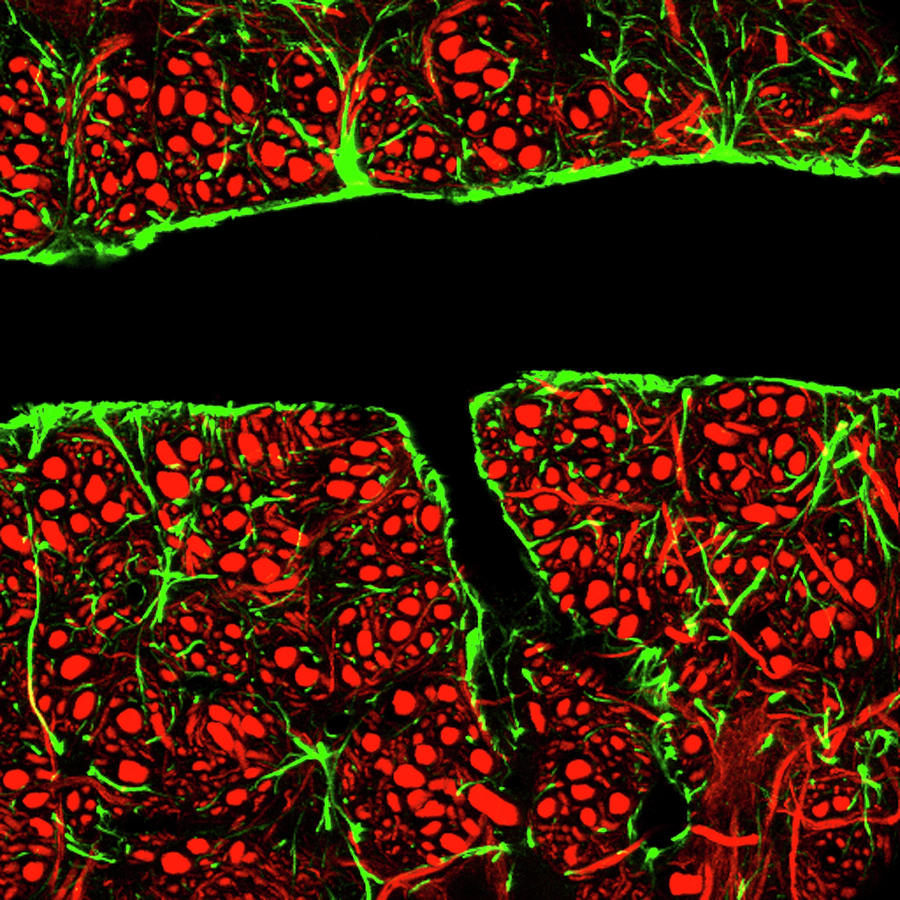
By testing the causal involvement of some of these using reverse genetics, we uncover a reinforcing role for angiopoietin 2 at the BBB. We also reveal heterogeneous modes of pericyte-deficient BBB impairment, where hotspot leakage sites display arteriolar-shifted identity and pinpoint putative BBB regulators. Adult Pdgfb ret/ret brains display ongoing angiogenic sprouting without concomitant cell proliferation providing unique insights into the endothelial tip cell transcriptome. We find that endothelial cells without pericyte contact retain a general BBB-specific gene expression profile, however, they acquire a venous-shifted molecular pattern and become transformed regarding the expression of numerous growth factors and regulatory proteins. We reveal transcriptional, morphological, and functional consequences of pericyte absence for brain endothelial cells using a combination of methodologies, including single-cell RNA sequencing, tracer analyses, and immunofluorescent detection of protein expression in pericyte-deficient adult Pdgfb ret/ret mice. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
